🧬 Partial Epigenetic Reprogramming Enters the Clinic
3/5/20261 min read
🧬 Partial Epigenetic Reprogramming Enters the Clinic
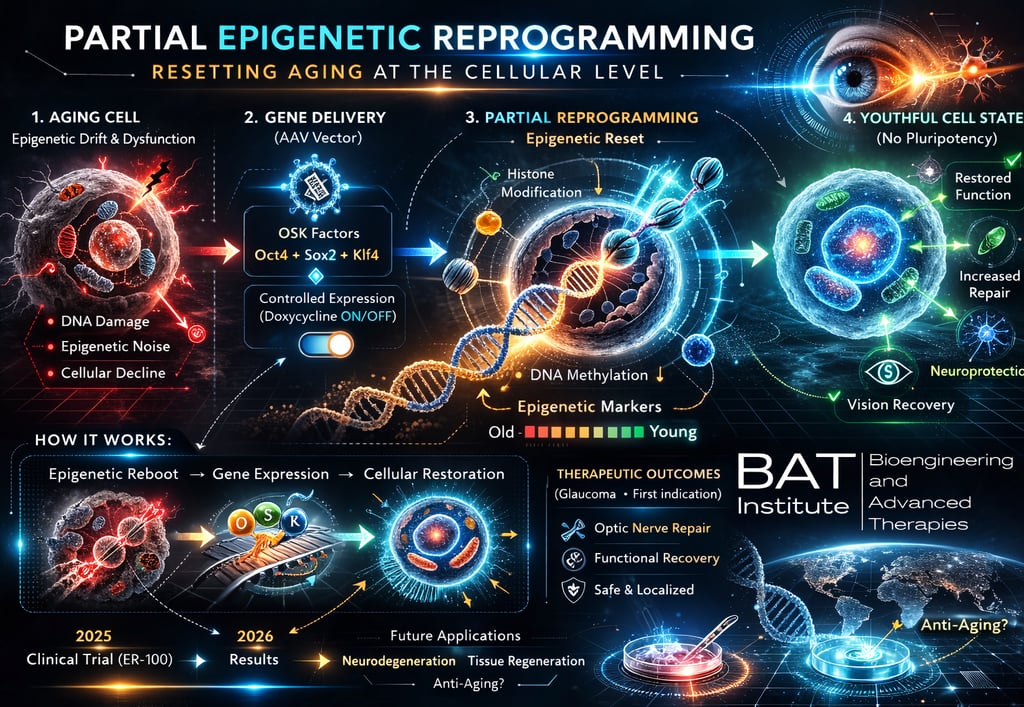
A first-in-human clinical trial is now underway to evaluate partial epigenetic reprogramming as a therapeutic strategy for age-related disease. The approach is being tested in glaucoma patients using the investigational gene therapy ER-100.
Technology overview
Partial epigenetic reprogramming is based on the transient expression of a subset of Yamanaka factors:
• Oct4
• Sox2
• Klf4
These factors aim to restore a more youthful epigenetic profile while maintaining cellular identity.
Mechanism of action
1️⃣ Aging-associated dysfunction
Cells accumulate epigenetic drift, altered DNA methylation patterns, and functional decline.
2️⃣ Targeted gene delivery
An AAV vector delivers OSK factors to retinal ganglion cells.
3️⃣ Controlled expression
Gene activity is regulated through an inducible system (doxycycline ON/OFF) to avoid full reprogramming.
4️⃣ Epigenetic reset
Partial remodeling of chromatin and DNA methylation patterns.
5️⃣ Functional recovery
Goal: restore cellular performance and support optic nerve function.
Why glaucoma as the first indication
• Localized delivery to the eye
• Immune-privileged environment
• Measurable functional endpoints
• Established clinical pathways for ocular gene therapy
Development pathway
Preclinical studies demonstrated restoration of optic nerve function in animal models.
The current clinical program is designed to evaluate safety, tolerability, and preliminary efficacy in humans.
Potential future applications
• Neurodegenerative diseases
• Tissue regeneration
• Age-related functional decline
Partial epigenetic reprogramming represents a new class of gene-based regenerative strategies now transitioning from preclinical research into human clinical evaluation.
More info: https://lnkd.in/emzDCEYW
hashtag#Biotech hashtag#GeneTherapy hashtag#Epigenetics hashtag#RegenerativeMedicine hashtag#longevity
