🚀 Beyond "Wear and Tear": How We Are Redefining Osteoarthritis and the regeneration of cartilage at the Molecular Level 🧬💡
5/7/20262 min read
🚀 Beyond "Wear and Tear": How We Are Redefining Osteoarthritis and the regeneration of cartilage at the Molecular Level 🧬💡
Interesting for those researchers working on articular cartilage regeneration. 🔬✨
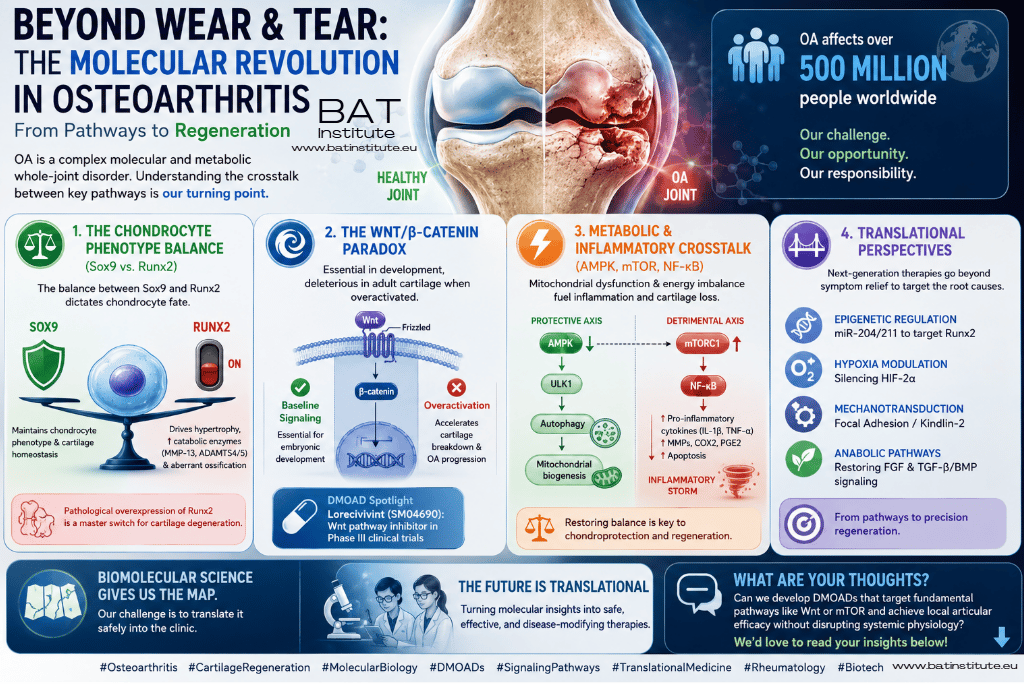
We are witnessing a drastic evolution in the paradigm of Osteoarthritis (OA): we no longer consider it a simple "wear-and-tear" mechanical condition, but rather a profoundly complex molecular and metabolic whole-joint disorder! 🧬🦴💥
As we analyze recent comprehensive literature (Chen et al., 2021 & Yao et al., 2023), we want to highlight the pivotal molecular mechanisms dictating how our chondrocytes proliferate, differentiate, and undergo apoptosis. These findings are actively redefining our current therapeutic targets: 🎯💊
📉 Our Balancing Act: The Chondrocyte Phenotype (Sox9 vs. Runx2) ⚖️
We know that cartilage homeostasis relies on the continuous expression of the transcription factor Sox9 to maintain the chondrocyte phenotype and block hypertrophic differentiation. 🛡️ Conversely, we observe that the pathological overexpression of Runx2 acts as a "master switch" 🎛️ triggering chondrocyte hypertrophy, promoting catabolic enzymes (MMP-13, ADAMTS4/5) ✂️, and driving our tissues toward aberrant endochondral ossification. 🦴
🚥 The Wnt/β-catenin Paradox We Must Navigate 🌪️
While we recognize that baseline canonical Wnt signaling is essential during embryonic development, we have found that the overactivation of the β-catenin axis in adult articular chondrocytes is highly deleterious and accelerates cartilage breakdown. 📉 This realization has driven us to develop Disease-Modifying Osteoarthritis Drugs (DMOADs), such as Lorecivivint (SM04690)—a Wnt pathway inhibitor that we are currently watching advance in phase III clinical trials! 🏥🚀
⚡ Our Metabolic and Inflammatory Crosstalk (AMPK, mTOR, and NF-κB) 🔋🔥
We increasingly understand that OA pathogenesis is intimately linked to mitochondrial dysfunction and energy imbalances. ⚡
• We see AMPK (our primary cellular energy sensor) showing severely reduced phosphorylation levels in OA, which suppresses ULK1-mediated autophagy 🧹 and impairs our mitochondrial biogenesis. 🛑
• Simultaneously, we recognize that the hyperactivation of mTORC1 actively suppresses protective autophagic mechanisms and exacerbates the NF-κB pathway 🚨. This orchestrates an inflammatory storm of pro-apoptotic and catabolic factors (MMPs, COX2, PGE2) within the joint. 🌪️🩸
🎯 Our Translational Perspectives 🌉
Our traditional pharmacological approach, strictly limited to palliative pain management, is facing a massive turning point. ⏳ We are now focusing our next-generation biological therapies on epigenetic regulators (like miR-204/211 to target Runx2) 🧬, silencing pathological hypoxic responses (HIF-2α) 🌬️, mechanotransduction modulators (Focal Adhesion/Kindlin-2) ⚙️, and restoring balance through our FGF and TGF-β/BMP pathways. 🌱
Understanding the deep crosstalk within these signaling cascades is our true turning point for achieving cartilage regeneration and treating a disease that disables over 500 million people globally. 🌍🤝
Biomolecular science is giving us the map 🗺️; our collective challenge now is safely translating these targeted gene and cellular therapies into clinical settings. 🏥👩🔬👨🔬
#Osteoarthritis 🦴 #CartilageRegeneration 🌱 #MolecularBiology 🧬 #TissueEngineering 🔬 #Chondrocytes 🦠 #DMOADs 💊 #SignalingPathways 🚥 #CellBiology 🧫 #TranslationalMedicine 🌉 #Biotech 🚀 #Rheumatology 🩺 #Orthopedics 🦵
References
Chen H, Tan X-N, Hu S, Liu R-Q, Peng L-H, Li Y-M and Wu P (2021) Molecular Mechanisms of Chondrocyte Proliferation and Differentiation. Front. Cell Dev. Biol. 9:664168. doi: 10.3389/fcell.2021.664168
Yao Q, Wu X, Tao C, Gong W, Chen M, Qu M, Zhong Y, He T, Chen S, Xiao G. Osteoarthritis: pathogenic signaling pathways and therapeutic targets. Signal Transduct Target Ther. 2023 Feb 3;8(1):56. doi: 10.1038/s41392-023-01330-w. PMID: 36737426; PMCID: PMC9898571.
